PCS general secretary FRAN HEATHCOTE explains why opposing war is inseparable from defending jobs, wages and public services – and why readers should come to the London Peace Conference on Saturday June 20
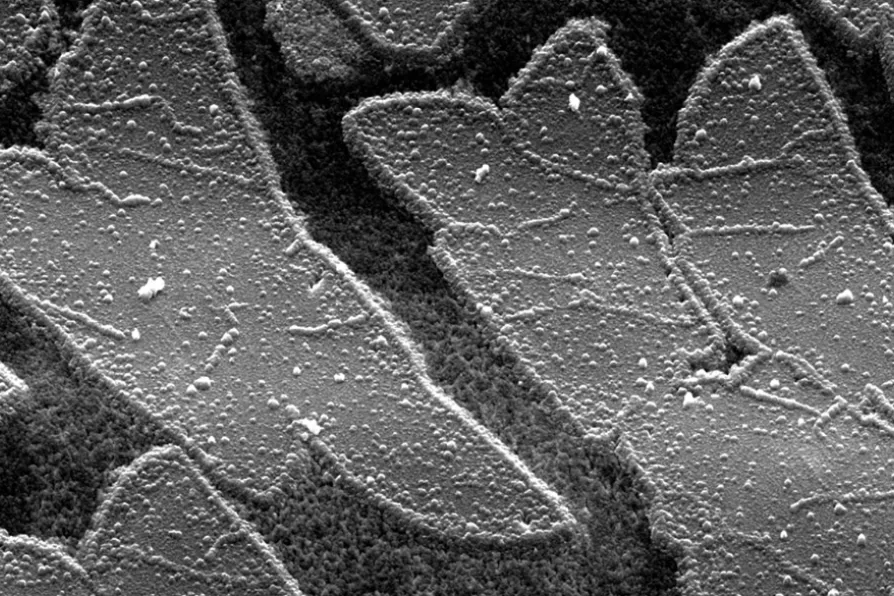
 These microscopic butterflies are one of the smallest, lightest and most expensive ones in the world as they partially consist of the novel one atom thick material graphene
[Maido Merisalu/Creative Commons]
These microscopic butterflies are one of the smallest, lightest and most expensive ones in the world as they partially consist of the novel one atom thick material graphene
[Maido Merisalu/Creative Commons]
EVERYTHING is made of atoms. To understand this, we’re asked to imagine that the smooth continuous materials around us are each made up of tiny lumps.
Rather than acting like little inert building bricks, each is humming with the energy of existing. Every atom is itself an immense void with even tinier positive and negative charged matter suspended in space, whizzing round each other and changing configuration to give the atom its own particular properties.
The dynamism of the charges moving around and interacting both inside the atom and outside of it defines the ways that charge and energy can move between them, through space, sometimes joining them together in configurations of molecules of infinite variety.

MIRANDA RICHMOND relishes the gloriously liberated art of Roy Oxlade, and traces his method back to the thinking of David Bomberg, his acknowledged teacher

Neutrinos are so abundant that 400 trillion pass through your body every second. ROX MIDDLETON, LIAM SHAW and MIRIAM GAUNTLETT explain how scientists are seeking to know more about them
New research into mutations in sperm helps us better understand why they occur, while debunking a few myths in the process, write ROX MIDDLETON, LIAM SHAW and MIRIAM GAUNTLETT

Olive oil remains a vital foundation of food, agriculture and society, storing power in the bonds of solidarity. Though Palestinians are under attack, they continue to press forward write ROX MIDDLETON, LIAM SHAW and MIRIAM GAUNTLETT









