PCS general secretary FRAN HEATHCOTE explains why opposing war is inseparable from defending jobs, wages and public services – and why readers should come to the London Peace Conference on Saturday June 20

IN THE coming weeks and months, the National Health Service will be tested to its limits, striving to save as many patients as possible who are seriously ill due to the coronavirus.
The surges in intensive-care patient numbers will cause major disruption to the routine work of the NHS, which includes crucial life-saving procedures.
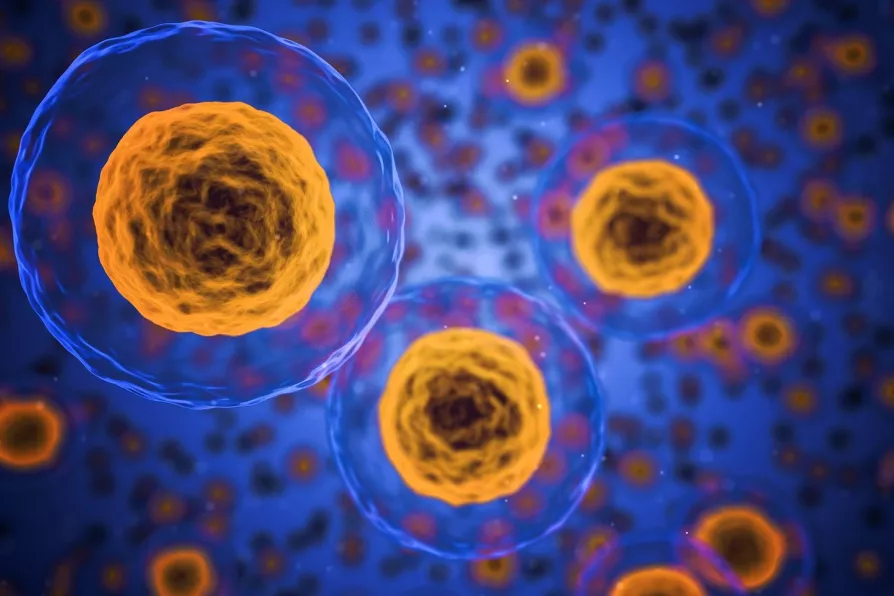
One such procedure that has been particularly affected by international travel disruption has been stem-cell donation, for the treatment of conditions such as blood cancer.
Once the donation has been made, the stem cells need to be transplanted into the recipient within 72 hours.
Our immune system uses special structures on the surfaces of cells called antigens to recognise cells that belong to our own body (“self”).
New research into mutations in sperm helps us better understand why they occur, while debunking a few myths in the process, write ROX MIDDLETON, LIAM SHAW and MIRIAM GAUNTLETT

Olive oil remains a vital foundation of food, agriculture and society, storing power in the bonds of solidarity. Though Palestinians are under attack, they continue to press forward write ROX MIDDLETON, LIAM SHAW and MIRIAM GAUNTLETT

ALEX DITTRICH hitches a ride on a jaw-dropping tour of the parasite world









